Clinical Trial CEB-01-RLS01-CT
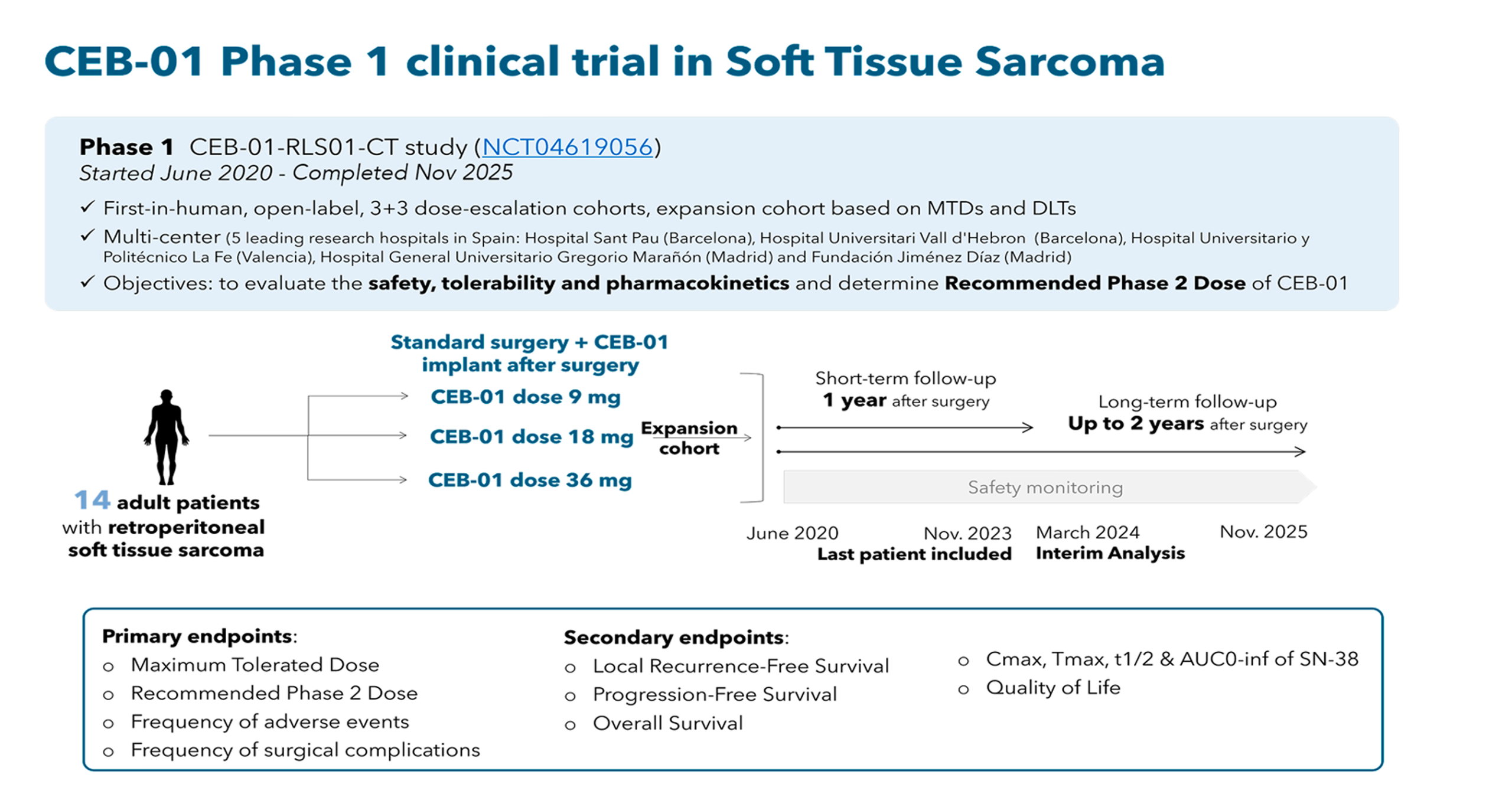
Phase 1, open-label, first-in-human clinical trial evaluating CEB-01 in adult patients with de novo or recurrent retroperitoneal soft tissue sarcoma, after surgery. The trial assessed safety, tolerability, pharmacokinetics, and preliminary efficacy of CEB-01 when used in addition to standard of care.
The trial enrolled 14 adult participants (≥ 18 years old) with histologically confirmed locally advanced (T3–T4) or recurrent retroperitoneal soft tissue sarcoma. Participants were allocated in a sequential dose-escalation design (3+3 modified Fibonacci method) across three cohorts to determine the recommended Phase 2 dose (RP2D) based on Maximum Tolerated Dose (MTD) (defined as < 33 % of patients experiencing dose-limiting toxicity within 2 weeks post-surgery):
Total SN-38 dose of 9 mg
Total SN3-8 dose of 18 mg
Total SN-38 dose of 36 mg
Trial sites (recruiting completed): Hospital Universitari Vall d’Hebron (Barcelona), Hospital de la Santa Creu i Sant Pau (Barcelona), Fundación Jiménez Díaz (Madrid), Hospital General Universitario Gregorio Marañón (Madrid), and Hospital Universitario y Politécnico La Fe (Valencia).
Recruitment period: Q2 2020 – Q4 2023
End of study: Q3 2025