CEB-01
Our lead clinical candidate, CEB-01, is a novel, biodegradable nanofiber membrane loaded with SN-38, the active metabolite of irinotecan. Placed directly at the tumor resection site, CEB-01 delivers a sustained release and a high local concentration of SN-38 over several weeks, enhancing postoperative tumor control and reducing recurrence without systemic toxicity. The CEB-01 treatment is an add-on to the standard of care and is compatible with current multi-modal therapeutic options for cancer.
Currrent Standard of care for non-metastatic cancers amenable to surgery
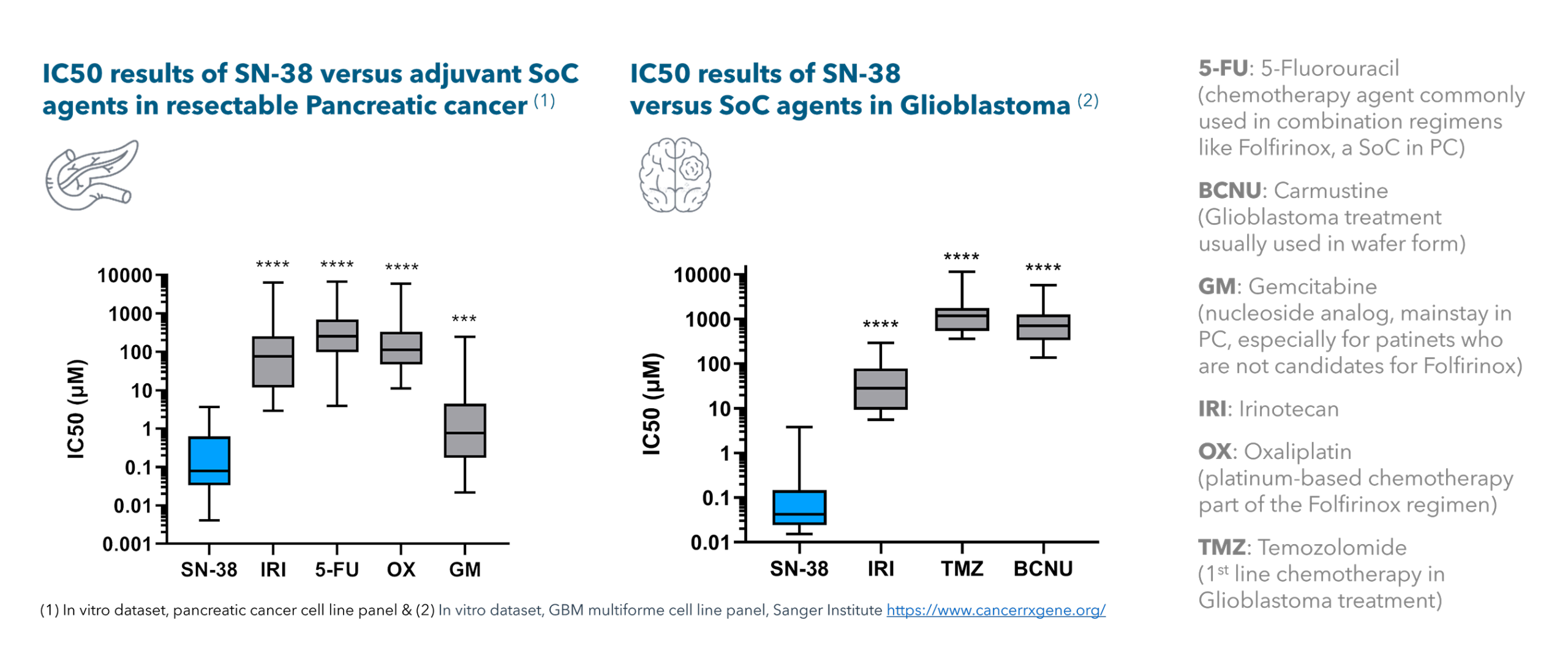
Preclinical evidence indicates that SN-38 exhibits strong cytotoxic activity against a variety of tumors, highlighting its potential as an effective anticancer agent. It shows particular potency in pancreatic cancer and glioblastoma, where it outperforms irinotecan (100 to 1000-fold more potent) and other standard-of-care therapies. In soft tissue sarcoma, SN-38-loaded membranes have been shown to provide a local, biocompatible, and effective drug delivery strategy. This approach efficiently damages tumor cells and leads to significantly improved in vivo survival compared to systemic or subcutaneous administration of irinotecan (Monterrubio et al., 2016).