Clinical Trial CEB-01-RLPC01-CT
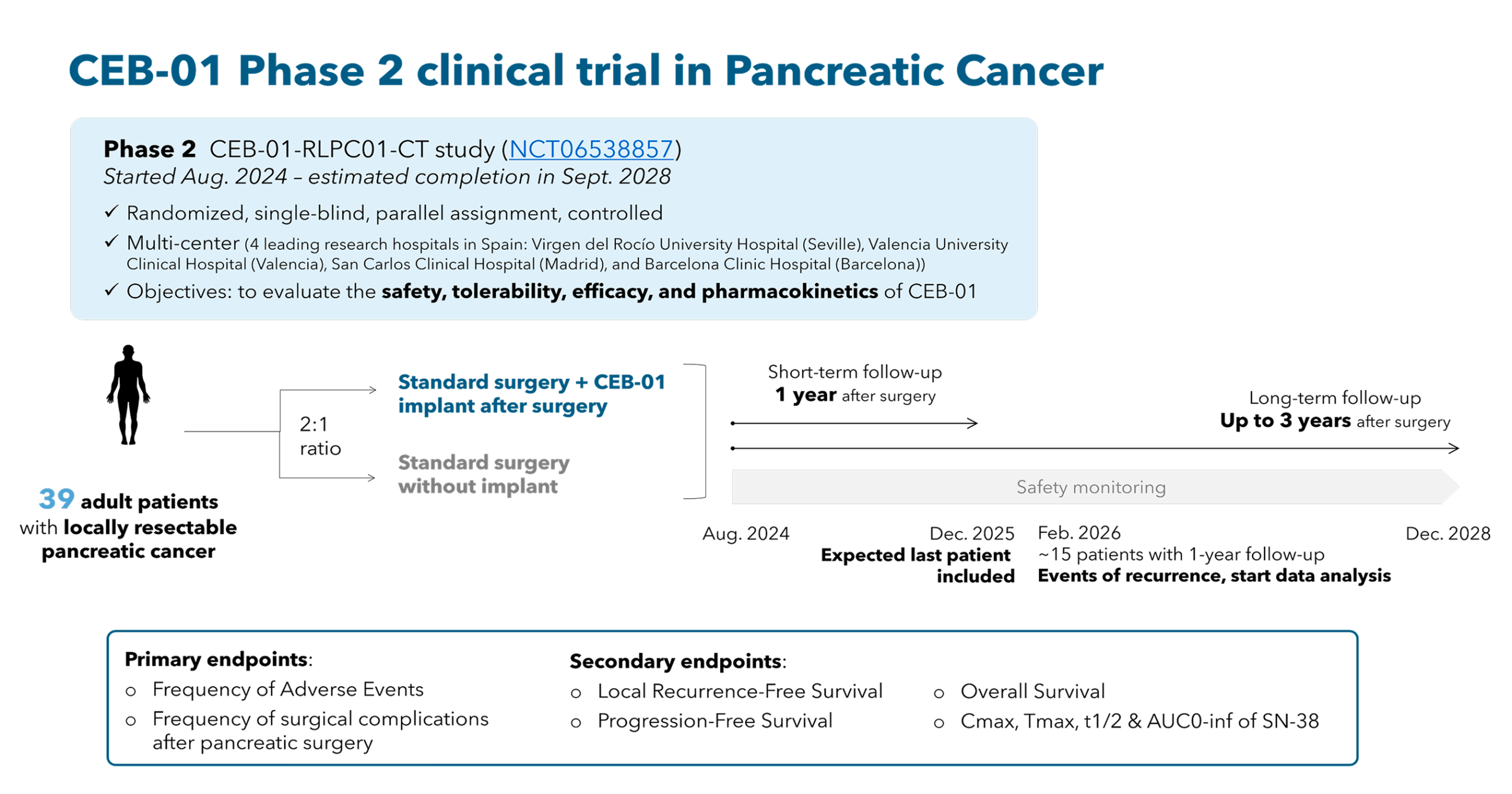
Phase 2, randomized, single blind, exploratory clinical trial evaluating CEB-01 in patients with de novo resectable pancreatic adenocarcinoma. The trial will assess the safety, tolerability, pharmacokinetics, and preliminary efficacy of CEB01 when added to standard surgical treatment and adjuvant therapy.
The trial will enroll 39 adult participants (≥ 18 years old) with histologically confirmed resectable or borderline resectable pancreatic adenocarcinoma. Participants will be randomized in a 2:1 ratio into two arms:
Standard surgery + CEB-01 implant (treatment)
Standard surgery alone (control)
The trial will include short-term (1 year) and long-term (up to 3 years) follow-up to monitor safety, surgical complications, local recurrence, progression-free survival (PFS), and overall survival (OS). Pharmacokinetic data on SN-38 will also be collected over approximately 60 days post-surgery.
Active recruiting sites: Hospital Virgen del Rocío (Sevilla), Hospital Clínic Barcelona (Barcelona), Hospital Clínico San Carlos (Madrid), Hospital Clínic de València (Valencia).
Recruitment period: Q3 2024 – Q2 2026
End of study: Q3 2028