News
First-in-human study of CEB-01 in retroperitoneal sarcoma
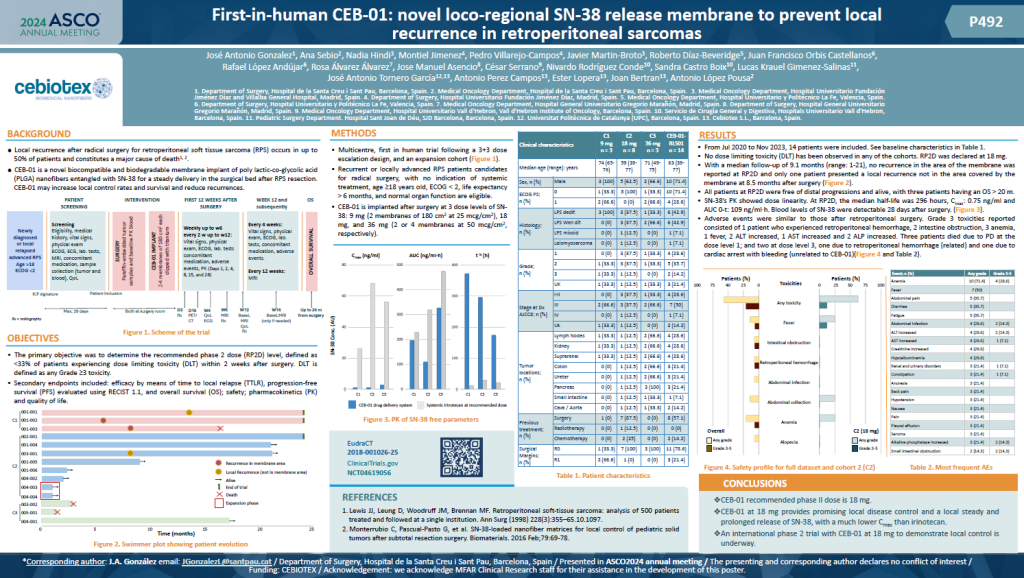
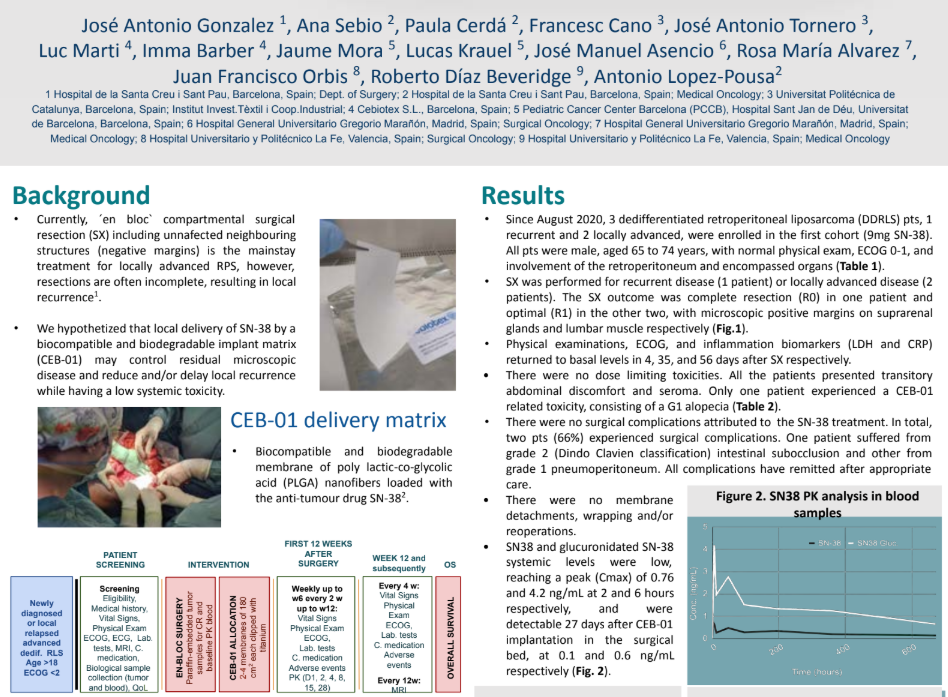
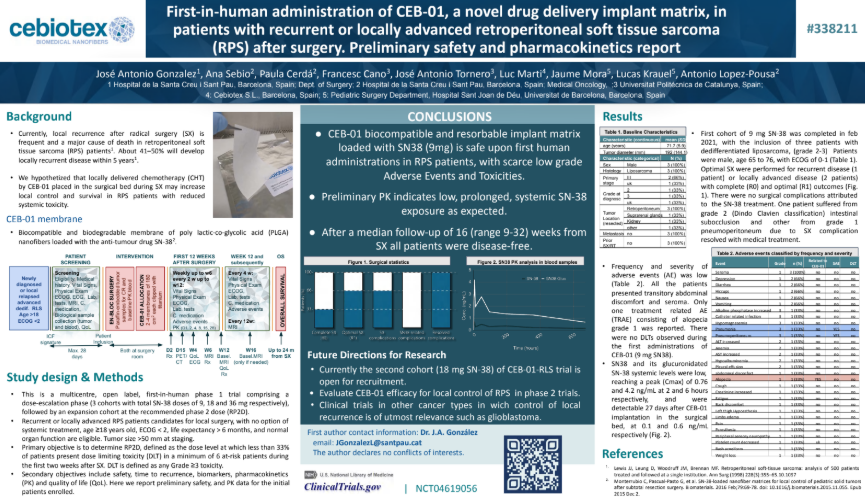
We are proud to announce the publication of our latest clinical results in Annals of surgical oncology, detailingthe first-in-human study of CEB-01, a novel biodegradable membrane for the local delivery of SN-38 aftertumour resection. This multicenter trial was designed to establish the recommended phase 2 dose (RP2D)while evaluating safety, efficacy, and pharmacokinetics. Key Highlights:✅ Positive […]
Latest news

First-in-human study of CEB-01 in retroperitoneal sarcoma
We are proud to announce the publication of our latest clinical results...

Cebiotex appoints Joan Torrejon as its new CEO
Cebiotex begins a new stage with a change in its executive management...

Cebiotex obtains EMA ODD for CEB-01 in Pancreatic Cancer
Cebiotex is proud to announce that CEB-01, our lead product for local...

CEBIOTEX receives approval to initiate a paediatric phase II clinical trial for locally resectable abdominal tumours
We are excited to share that the clinical trial to evaluate the...

CEBIOTEX achieves a key milestone: the launch of in-house GMP-certified manufacturing facilities to support the production of CEB-01
𝗖𝗘𝗕𝗜𝗢𝗧𝗘𝗫 𝗮𝗰𝗵𝗶𝗲𝘃𝗲𝘀 𝗮 𝗸𝗲𝘆 𝗺𝗶𝗹𝗲𝘀𝘁𝗼𝗻𝗲: 𝘁𝗵𝗲 𝗹𝗮𝘂𝗻𝗰𝗵 𝗼𝗳 𝗶𝗻-𝗵𝗼𝘂𝘀𝗲 𝗚𝗠𝗣-𝗰𝗲𝗿𝘁𝗶𝗳𝗶𝗲𝗱 𝗺𝗮𝗻𝘂𝗳𝗮𝗰𝘁𝘂𝗿𝗶𝗻𝗴 𝗳𝗮𝗰𝗶𝗹𝗶𝘁𝗶𝗲𝘀...
Launch of First Pancreatic Cancer Clinical Trial with CEB-01 in Spain
We are pleased to announce our first clinical trial in pancreatic cancer,...